How the media’s homophobic and racist coverage of monkeypox mirrors the HIV/AIDS epidemic
As of 21 May 2022, the World Health Organization (WHO) received reports of 92 laboratory-confirmed monkeypox cases and 28 suspected cases from 12 countries where the disease is not usually found, including several European nations, the US, Australia and Canada.
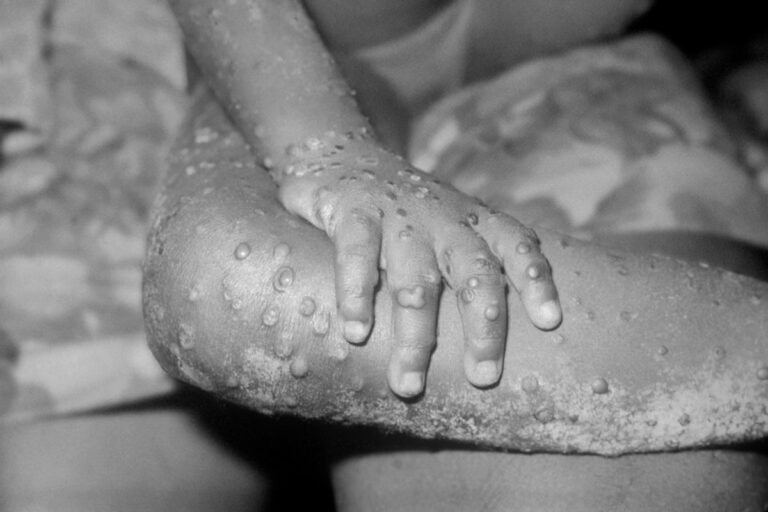
Monkeypox is a virus transmitted to humans from animals—also called a viral zoonosis—with symptoms very similar to those seen in past smallpox patients (although much less severe). Some of its symptoms include having: fevers, intense headaches, swelling of the lymph nodes, back pain, muscle aches and, more notoriously, skin eruptions with rashes concentrated on the face and extremities of someone infected.
The rash evolves from macules (lesions with a flat base) to papules (slightly raised, firm lesions), vesicles (lesions filled with clear fluid), pustules (lesions filled with yellowish fluid) and crusts which subsequently dry up and fall off. The number of lesions can vary from a mere few to several thousand, but in severe cases, they can even coalesce until large sections of skin slough off.
Though some of the monkeypox symptoms listed above can seem quite scary, especially with visuals, the disease is usually a self-limited illness with the symptoms lasting from two to four weeks. According to the WHO, the case fatality ratio of the virus has historically ranged from 0 to 11 per cent in the general population and has been higher among young children. In recent times, the case fatality ratio has been around 3 to 6 per cent.
Monkeypox primarily occurs in Central and West Africa, often in proximity to tropical rainforests, though it has been increasingly appearing in urban areas. The first human case was identified in a child in the Democratic Republic of the Congo in 1970. Animal hosts include a range of rodents and non-human primates.
The virus is transmitted from one person to another by close contact with lesions, body fluids, respiratory droplets and contaminated materials such as bedding. Transmission via droplet respiratory particles usually requires prolonged face-to-face contact, which puts health workers, household members and other close contacts of active cases at greater risk.
But just like we didn’t have much information on the virus when COVID-19 first spread, the WHO highlighted that “while close physical contact is a well-known risk factor for transmission, it is unclear at this time if monkeypox can be transmitted specifically through sexual transmission routes.” In other words, studies are needed to better understand this risk.
Regardless of the current lack of knowledge and information we have on monkeypox so far, this hasn’t stopped many news outlets from portraying the virus as highly transmissible among LGBTQIA+ and African individuals. Just shy of a month since the recent spread was first identified, news reports on monkeypox have been denounced as racist and homophobic. The worrying approach to the coverage of the disease will only exacerbate stigma and undermine the global response to the growing outbreak, the United Nations’ Aids agency (UNAIDS) wrote in a press release.
Instead, UNAIDS said that although “a significant proportion” of recent monkeypox cases have been identified among gay, bisexual and other men who have sex with men, transmission most likely happened via close physical contact with a monkeypox sufferer, which means it could affect anyone.
“Stigma and blame undermine trust and capacity to respond effectively during outbreaks like this one,” said the UNAIDS deputy executive director, Matthew Kavanagh. “Experience shows that stigmatising rhetoric can quickly disable evidence-based response by stoking cycles of fear, driving people away from health services, impeding efforts to identify cases and encouraging ineffective, punitive measures,” he continued.
Meanwhile, on Channel 4’s TikTok page, @C4news, the news platform got into hot water for a recently posted video that focused on the spread of the disease. In it, the presenter can be heard saying, “Several of the people infected are gay and bisexual men.” Though the video goes on to state that the virus is transmitted through skin-on-skin contact, simply starting with a highlight on the sexuality of those who have already been infected will negatively impact the LGBTQIA+ community, both internally and externally.
@c4news Monkeypox cases are rising in the UK - but how dangerous is it? #news #monkeypox #uk #c4news #channel4news #learnontiktok
♬ original sound - Channel 4 News
This worrying and discriminatory trend in reporting can easily be compared to the AIDS epidemic, in which the media played a very powerful role in shaping the public’s perception of the condition caused by the HIV virus. It is because of such biased reporting at the start of the AIDS/HIV epidemic that masses of the population believed (and for some, still believe) it to be a ‘gay virus only’. Of course, this claim was medically untrue and only served to perpetuate harmful, dangerous stereotypes about people living with HIV and members of the LGBTQIA+ community—especially black and brown queer people.
The same harmful tactics seem to be reemerging in the coverage of this latest disease. If we’re not careful, we might just witness history repeating itself. That being said, it should also be noted that this time, we have hindsight on our side—making us better able to publicly voice and challenge the archaic homophobic and racist tropes still evident in traditional media.